Click Here to Download this Blog Post – The Overactive Guard Dog: Understanding Mast Cell Activation Syndrome (MCAS) (Part 14)
By Dr. Nicholas L. DePace, M..D., F.A.C.C – Cardiologist specializing in autonomic dysfunction, Ehlers-Danlos syndrome and POTS.
- If you have hEDS or HSD, you might feel like you are allergic to the world. You aren’t just imagining it. Many people with connective tissue disorders also battle an immune system issue called Mast Cell Activation Syndrome (MCAS).
What are Mast Cells?
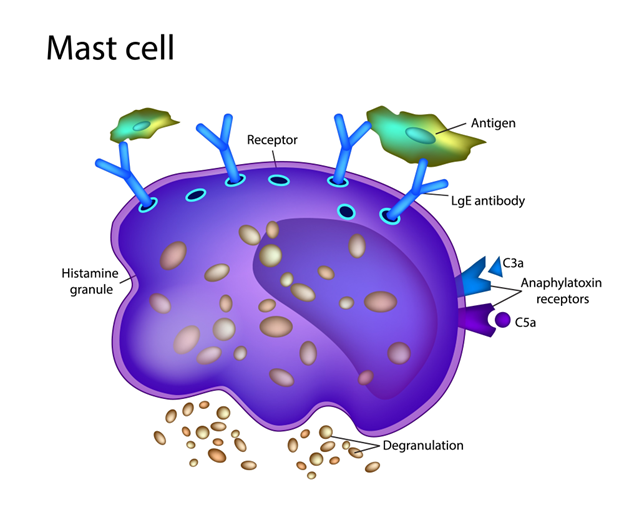
Think of mast cells as your body’s “guard dogs.” They live in almost every tissue and protect you from invaders like bacteria and viruses. When they spot a threat, the Sympathetic Nervous System (the gas pedal) tells them to attack by releasing chemicals like histamine.
When the Guard Dog Goes Rogue
In a healthy body, the Parasympathetic Nervous System (the brake pedal) keeps these cells calm. But because hEDS/HSD causes these nerves to glitch, the “brake” fails.
The result? Your mast cells become confused. They start barking and biting at things that aren’t dangerous—like foods, smells, or temperature changes. This overreaction is MCAS.
The Symptoms
Because mast cells are everywhere, the symptoms can happen anywhere. It’s not just sneezing; it often looks like:
- Skin:Hives, flushing, itching.
- Gut:Nausea, vomiting, diarrhea (often misdiagnosed as IBS).
- Heart:Racing heart, low blood pressure, fainting.
- Lungs:Wheezing or throat tightness.
- Brain:Brain fog and headaches.
How is it Diagnosed?
Doctors look for the “Rule of Three”:
- Clinical Signs:Symptoms in two or more body systems (e.g., skin and stomach).
- Lab Tests:High levels of specific chemicals (like Tryptase or histamine) in blood or urine. Note: You might also be tested for a genetic trait called HaT (Hereditary Alpha-Tryptasemia), which is common in EDS patients.
- Response to Treatment:You feel better when taking mast cell medications.
The “Two-Door” Treatment Strategy
Treating MCAS isn’t just about taking an allergy pill. Histamine has different ways of entering your cells, like different doors. To stop the reaction, you often need to lock both doors:
- Block Door 1 (H1 Blockers):Common allergy meds like Zyrtec or Xyzal.
- Block Door 2 (H2 Blockers):Meds often used for acid reflux, like Pepcid.
If you only block one, the body just uses the other door, and you don’t get relief.
Where to Seek Expert Care?
It is important to seek out a clinician with expertise in EDS to make an accurate diagnosis and create a treatment plan. One of the nation’s leading centers is Franklin Cardiovascular Associates, under the direction of Nicholas DePace, MD, FACC. They are located in Sicklerville, New Jersey. franklincardiovascular.com, (856) 589-6034
About the Author
Nicholas L. DePace, MD, FACC is a board-certified cardiologist and Medical Director of Franklin Cardiovascular Associates. A graduate of the Mount Sinai School of Medicine, Dr. DePace has decades of clinical, academic, and research experience and has held faculty appointments as a Clinical Professor of Medicine, becoming one of the youngest full professors in Philadelphia at the time of his appointment.
Dr. DePace specializes in the diagnosis and treatment of autonomic nervous system dysfunction (dysautonomia), including POTS, autonomic dysfunction associated with Ehlers-Danlos syndrome (EDS), chronic fatigue, and anxiety-like conditions that are frequently misdiagnosed. He is nationally recognized for his work on parasympathetic and sympathetic (P&S) nervous system imbalance, a core mechanism underlying many complex chronic disorders.
In addition to treating patients from across the United States, Dr. DePace is a prolific clinical researcher and author of multiple nationally distributed medical textbooks published by Springer and W.W. Norton, focusing on autonomic dysfunction, mitochondrial disorders, cardiovascular disease, and mind–body medicine.
👉 View Dr. DePace’s professional profile
👉 View medical books by Dr. DePace